
Protecting Pregnant Women’s Cardiovascular Health
Pemi31 Therapeutics is an innovative Women’s Health Biotech Company that is preparing to initiate preclinical studies that would support an IND filing and lead to a Phase 1 Clinical Trial. The novel intervention is Heat Shock Protein 27 Immunotherapy that, in preclinical studies, effectively reduces levels of cholesterol and inflammation. This groundbreaking therapy would harness natural proteins for the prevention of coronary artery disease (CAD) in Women of Childbearing Age (WCBA) with specific high-risk cholesterol disorders. Currently, there are no cholesterol-lowering therapies that are approved for pregnant or breastfeeding women. Going forward, we will explore an Orphan Drug Development Strategy to bring this transformative therapy to those in need.
Orphan Drug Development Opportunity:
Treatment of WCBA with (both) Familial Hypercholesterolemia [FH] and
elevated Lipoprotein(a) Levels [Lp(a)]
Most WCBA are relatively healthy and low risk for cardiovascular disease. However, there are unique populations of women with markedly elevated risk of coronary artery disease, who can ill-afford to delay preventative lipid-lowering therapies in order to avoid cardiovascular events such as heart attacks. For example:
Approximately 30% of patients with FH also have elevated Lp(a) levels and have a 5-fold increase in premature Coronary Artery Disease (CAD)
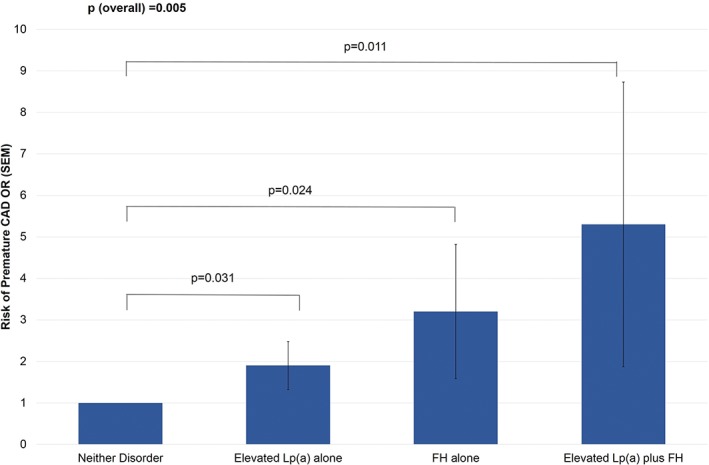
KL Ellis et al. Clinical Cardiology. 2018;41:378–384.
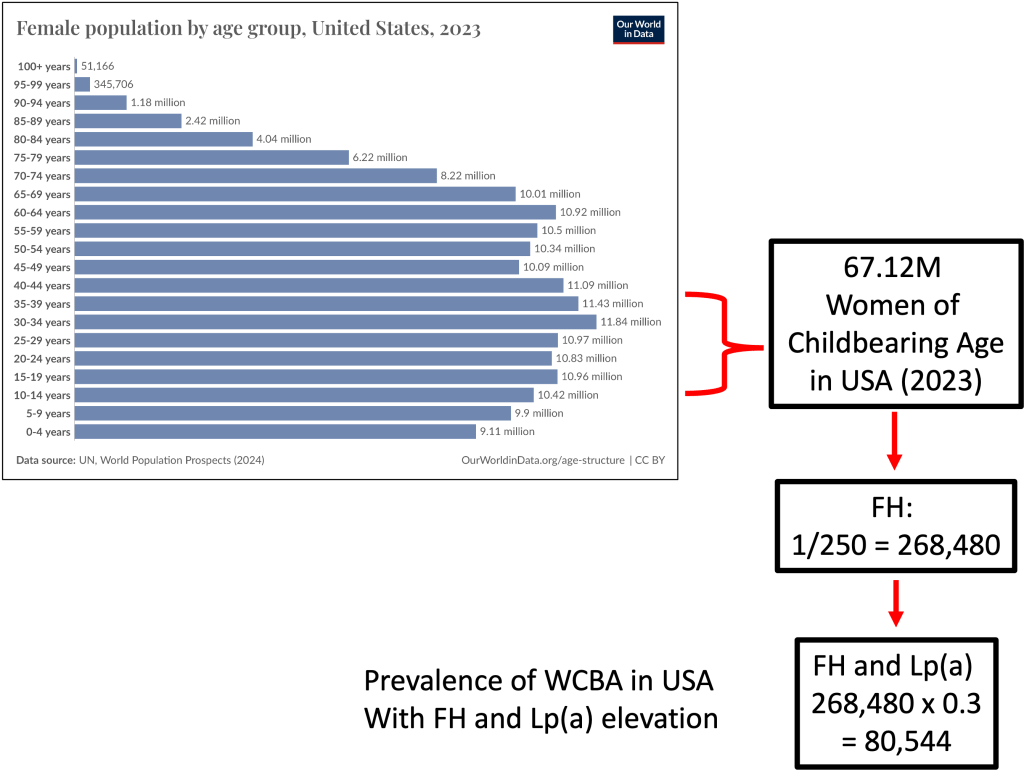
Prevalence of WCBA in USA with FH and Lp(a) Elevation
Critical Unmet Need:
Currently there are no cholesterol lowering therapies that are approved for pregnant or breast feeding women
Unique Value Proposition:
- Due to safety concerns, WCBA cannot take conventional cholesterol lowering medications (e.g., statins, ezetimibe, PCSK9 inhibitors) nor newer medications that are being developed to lower Lp(a) levels during pregnancy and while breast feeding.
- In the absence of any lipoprotein lowering therapy these WCBA are at high cardiovascular risk during pregnancy and while breast feeding.
- Hence, there are potential devastating outcomes for both the mother and the baby.
Therapeutic Advantage of Pemi31 Therapeutics Vaccination Therapy
- Vaccination with Heat Shock Protein 27 (an endogenous protein) is anticipated to have a clean safety profile – like other vaccines used during pregnancy, including Tdap (tetanus, diphtheria, pertussis). In contrast, “live” vaccines that contain weakened but still living forms of viruses or bacteria (such as measles, mumps and rubella) are contra-indicated during pregnancy.
- HSP27 Vaccination could lower LDL cholesterol and inflammation during pregnancy and while breast feeding.
- Hence, there is the potential to prevent acute cardiovascular events in WCBA with high CAD risk
Market Differentiation
This is a specific population of under 100,000 patients and therefore would qualify as an Orphan Drug Development program with:
- Financial Incentives:
- Tax credits for clinical research costs (up to 25% in the US)
- Waived FDA user fees (saving millions in application costs)
- Federal research grants
- Market exclusivity (7 years in US, 10 years in EU) protecting from competition
- Regulatory Benefits:
- Expedited review process
- More frequent FDA interactions and guidance
- Smaller clinical trials often accepted due to limited patient populations
- Flexibility in trial design and endpoints
- Reduced evidence requirements in some cases, given urgency of unmet needs
- Commercial Advantages:
- Premium pricing potential due to limited competition and small patient population
- Enhanced patient access through specialized distribution networks
- Stronger relationships with patient advocacy groups
- Possibility to expand to other indications later
- Development Efficiency:
- Faster path to market (average 2.3 years shorter than traditional drugs)
- Lower patient recruitment costs for trials
- Reduced marketing expenses due to targeted specialist physician base
- Potential to leverage natural history studies instead of placebo controls
The Discovery
While studying how estrogen affects blood vessels, we found that Heat Shock Protein 27 (HSP27) plays a crucial role in preventing atherosclerosis. Our research indicated that natural antibodies to HSP27 enhance its effectiveness in managing inflammation and cholesterol in the liver.
To explore the potential of HSP27 antibodies in reducing atherosclerosis, we tested a vaccination strategy. Animal studies revealed that HSP27 vaccination boosted antibodies and increased liver receptors (LDL-R) that clear cholesterol from the blood, while also reducing inflammation via the TLR4 pathway. This dual approach could effectively combat the development of coronary artery disease (CAD).
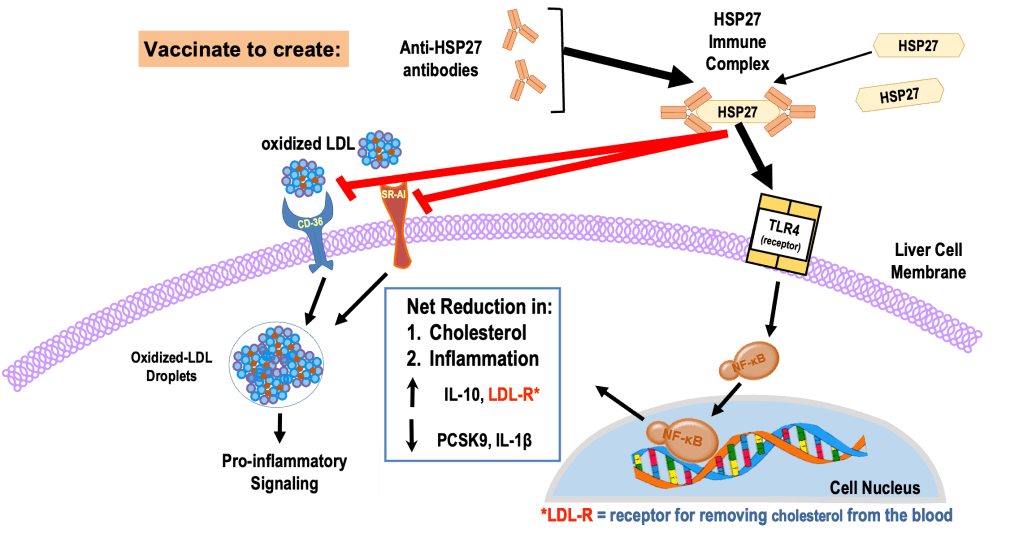
HSP27 Immune Therapies:
Mimicking the Health Benefits of Natural Proteins